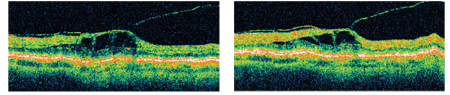
This study was performed in accordance with the ethical standards of the Declaration of Helsinki and was approved by the institutional review board of Inha University Hospital.Īll 85 enrolled patients had received complete monthly ophthalmic evaluations, which included best corrected VA, fluorescein angiography, and SD-OCT. We treated if there was an increase in OCT ≥100 µm, a loss of vision ≥1 line, or if a de novo macular hemorrhage was evident by clinical examination. In brief, further treatment was performed if intraretinal fluid (IRF) or subretinal fluid (SRF) persisted or recurred regardless of the presence of pigment epithelial detachment (PED). After three consecutive monthly loading doses, the fourth monthly treatment was determined according to the criteria recommended by the PrONTO and SUSTAIN studies. Subjects with disease duration less than one month and those who were treated with intravitreal ranibizumab (0.50 mg) for at least three months were selected. We retrospectively reviewed the records of 85 treatment-nave patients newly diagnosed with choroidal neovascularization (CNV) secondary to AMD at the vitreoretinal clinic of Inha University Hospital. Furthermore, it has been suggested that vitreomacular adhesion (VMA) and incomplete PVD-related traction force on the fovea contribute to the development and progression of AMD. Although AMD primarily involves outer retinal layers, several authors have suggested that the vitreous plays a role in the pathogenesis and/or progression of AMD, especially in the presence of incomplete posterior vitreous detachment (PVD) and its associated vitreomacular traction (VMT). 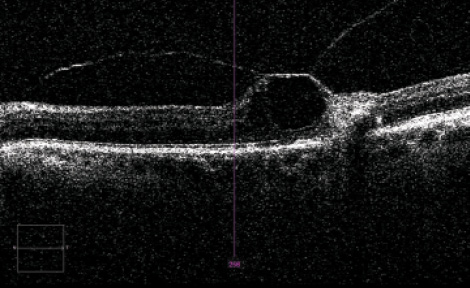
Additionally, it is important to identify the factors that correlate to OCT/VA outcome in AMD patients receiving customized PRN anti-VEGF treatment. Optical coherence tomography (OCT)/visual acuity (VA)-guided, individualized pro re nata (PRN) treatment, after three initial loading doses, is now regarded to be a successful and cost-effective option to monthly treatment, as verified by several studies. Ranibizumab (Lucentis Novartis Pharma AG, Basel, Switzerland Genentech Inc., San Francisco, CA, USA) is a humanized monoclonal antibody fragment targeting multiple isoforms of human vascular endothelial growth factor (VEGF)-A and has become a standard treatment for neovascular age-related macular degeneration (AMD).
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
